Table of Contents
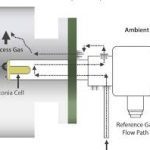
- Zirconia oxygen analyzer is specially used to measure the net concentration of oxygen in flue gas. It measures % of O2 by reading the voltage developed across a heated (750deg. Celsius) zirconium disc cell. When operated at proper temperature, the MV output of the cell is given by equation
E.M.F. (in mV) = KT log P1/P2 +C
P2 is the partial pressure of oxygen in the flue gas at one side of the cell,
P1 is the partial pressure of oxygen in the reference gas on other side of the cell. Instrument air is used as a reference gas.
T is the absolute temperature,
C is the cell constant and
K is the arithmetic constant.
2. When the cell is at proper operating temperature and there are unequal oxygen concentration across the cell, O2 ions will travel from the high partial pressure of O2 to the low partial pressure side of the cell. Because the output signal is proportional to the log of the inverse of the sample O2 partial pressure, the output signal decreases as the O2 concentration increases. The resulting logarithms output voltage is approx.53MV/decade.
3. A ceramic filter is provided at the tip of the O2 sensor probe to avoid the inrush of carbon and dust particles in the sensor.

Zirconium oxide is a trusted, industry-proven sensing technology for fast and accurate oxygen analysis in a wide range of applications, particularly combustion processes.
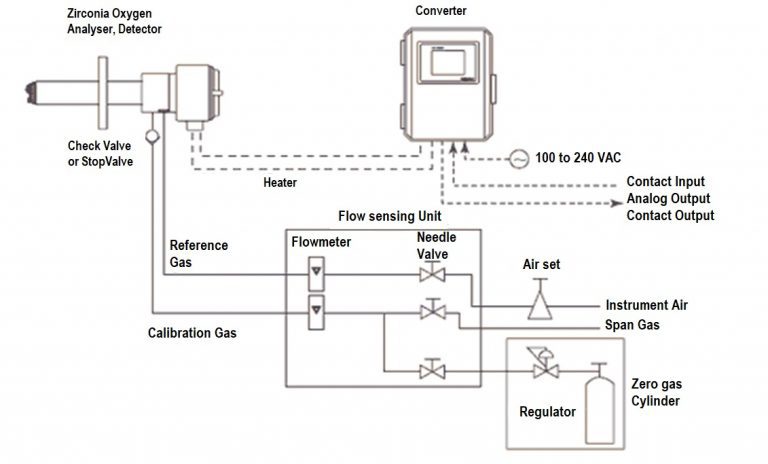
The Electronic Converter
- The converter has a temperature controller, amplifier and power supply card.
- The electronic converter converts the MV output signal into 4-20 MA signal which can be used as a indication.
- It also controls the temperature of zirconia sensor about (750deg. Celsius) and gives the digital display of % of O2.
- The amplifier receives MV signal generated by the sensor and produces a standard 4-20 Ma output signal.
Schematic Diagram of O2 Analyser




Read Also:-
How Infra Red(IR) Gas analyzer Work
Related Search:-





Quick Exhaust Valve
Quick Exhaust Valve





Tuning fork Type Level Switch
Tuning fork Type Level Switch





Temperature Switches
Temperature Switches




How To Zirconia oxygen analyzer works
How To Zirconia oxygen analyzer works





Paramagnetic Type Oxygen analyzer
Paramagnetic Type Oxygen analyzer





Displacer type level switch working principle
Displacer type level switch working principle